Please listen to this interview in which Eric, also known as FM8, a firefighter/emergency medical technician with 18 years of experience, talks about his fight and his experiences with vaccinated and BLE (Bluetooth low energy)
https://stephandubeau.substack.com/p/stephan-d-interviews-fm8-part-3
https://firemedic8.substack.com/p/exposing-suffolk-part-one-the-chief
BLE (Bluetooth low energy) …
https://patents.justia.com/patent/11893567
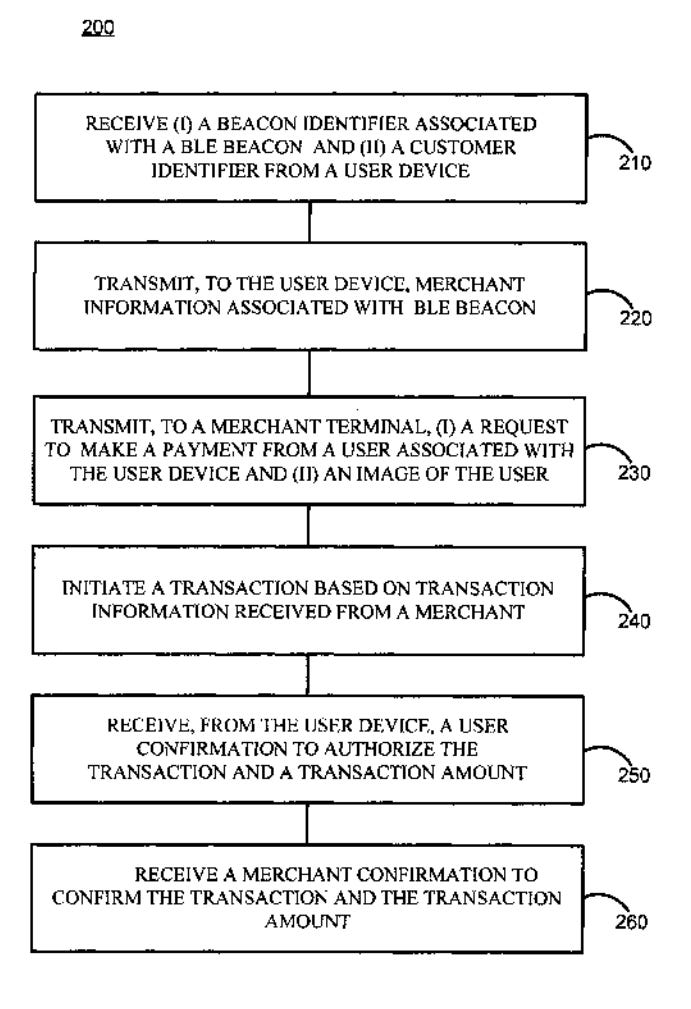
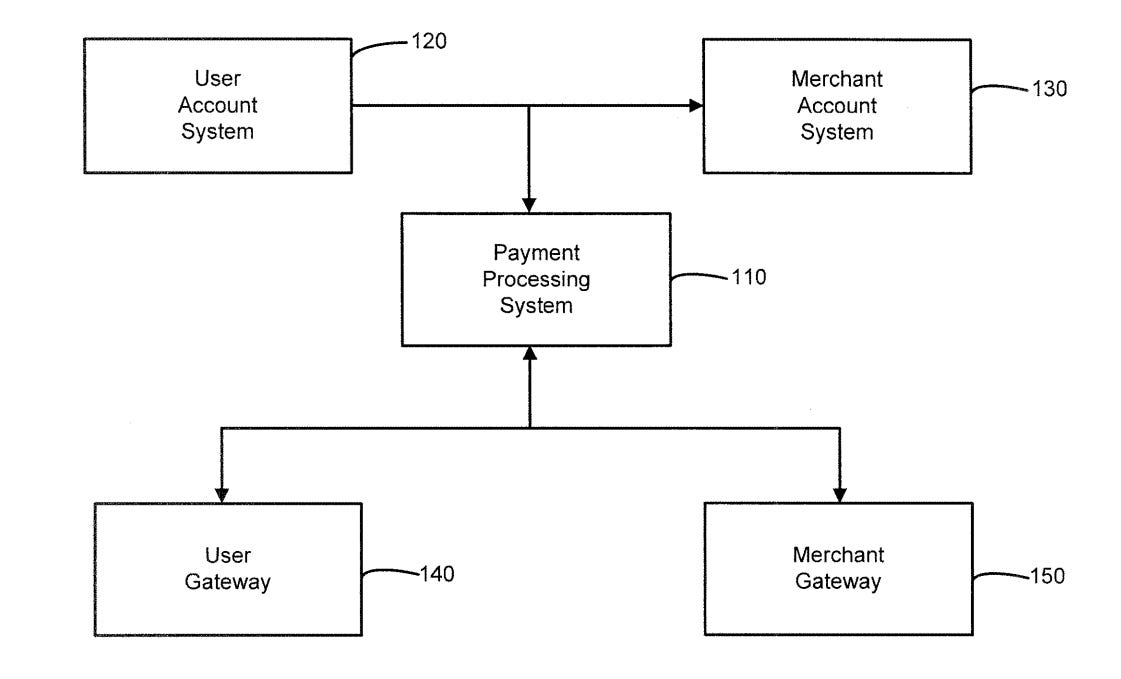
System and method for providing a bluetooth low energy mobile payment system
May 17, 2021 - American Express Travel Related Services Company, Inc.
A BLUETOOTH low energy mobile payments system may comprise a BLE beacon that is deployed at a merchant location. The BLE beacon may be associated with a specific merchant terminal. The BLE beacon may be configured to broadcast a unique beacon ID that is detectable by a user device. The user device may be configured to communicate the beacon ID to a payment system In response to receiving the beacon ID, the user device may initiate a payment between a user and a merchant, via a user device and a merchant terminal.
Description
CROSS REFERENCE TO RELATED APPLICATIONS
This application is a continuation of, claims priority to and the benefit of, U.S. Ser. No. 16/109,046, filed Aug. 22, 2018, entitled “TRANSACTIONS USING A BLUETOOTH LOW ENERGY BEACON,” which is a continuation of U.S. Pat. No. 10,062,073, issued Aug. 28, 2018 and filed Aug. 26, 2014, entitled “SYSTEM AND METHOD FOR PROVIDING A BLUETOOTH LOW ENERGY MOBILE PAYMENT SYSTEM,” the contents of which are incorporated herein by reference in their entirety for all purposes.
FIELD
The present disclosure relates to facilitating mobile payments, and more specifically, to systems and methods for creating a mobile payment system using BLUETOOTH low energy beacons.
https://patentimages.storage.googleapis.com/01/ab/56/98febd7b824eae/US10062073.pdf System and method for providing a BLUETOOTH low energy mobile payment system
https://novelbits.io/bluetooth-low-energy-ble-complete-guide/
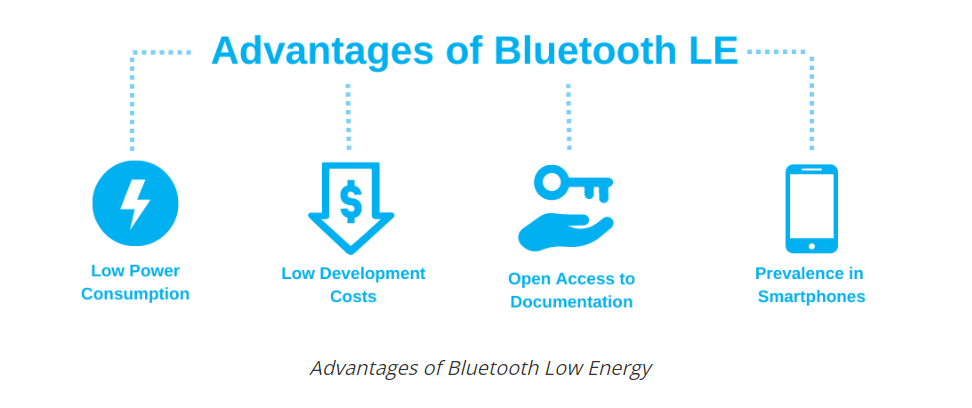
The Internet of Things (IoT) has been a buzzword for decades, but today this market is far from theoretical. There are over 10 billion active IoT devices globally, and one of the key technologies enabling this market growth is Bluetooth Low Energy (BLE).
Introduced in 2012, this flexible protocol provides wireless connection for many of IoT’s most critical applications. Its focus on operating with ultra-low power allows it to be used across industries such as consumer electronics, healthcare, and logistics.
What is Bluetooth Low Energy (BLE)?
Despite taking many cues from its predecessor, Bluetooth Classic, Bluetooth Low Energy is regarded as a different technology that specifically targets markets where the demand is for ultra-low power rather than high throughput.
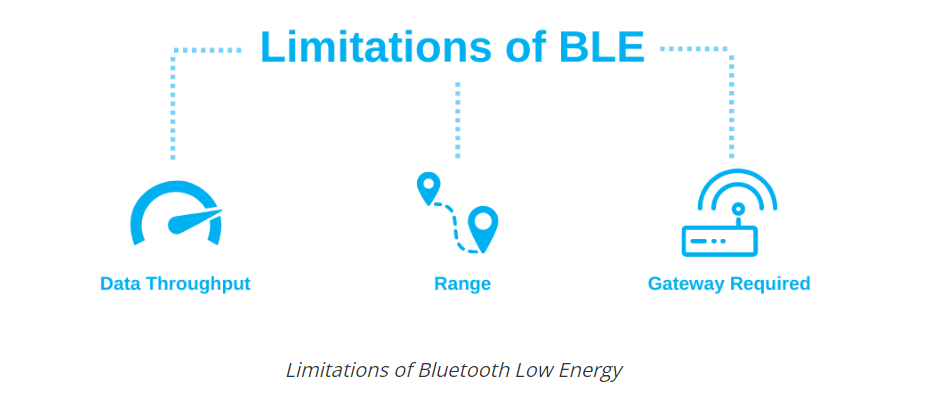
Data communication with an LE radio happens in short bursts that do not need to be very frequent. A typical LE use case would include periodically turning on the radio, transferring or receiving a few bytes or kilobytes of data, and then turning off and going back to sleep.
BLE was designed for short-range applications; hence, its range of operation is limited. There are a few factors that limit the range of BLE, including:
It operates in the 2.4 GHz ISM spectrum, which is greatly affected by obstacles that exist all around us, such as metal objects, walls, and water (especially human bodies).
Performance and design of the antenna of the BLE device.
The physical enclosure of the Bluetooth Low Energy device affects the antenna performance, especially
if it is an internal antenna.
Device orientation – effectively relates to the positioning of the antenna (e.g., in smartphones).
To transfer data from a BLE-only device to the Internet, another BLE device that has an IP connection is needed to receive this data and then, in turn, relay it to another IP device (or to the internet).
Contact tracing: Smart contact tracing systems are starting to emerge to help prevent the spread of infectious diseases. Instead of manually reporting data that can be used to identify those who have been in close contact with an infected person, BLE-enabled contact tracing allows for continuous scanning of BLE “tags” or smartphones to anonymously track human touchpoints.
Item finding tags: An increase in travel chaos has had some people adding AirTags or Tiles to their luggage in case it gets lost. These
BLE-enabled precision tracking devices
can be used for more than just luggage, though. You can pop one on anything you think you might lose, like a bicycle or your car keys, and track its location via a smartphone app.
Targeted ads: Imagine being at the clothing store and getting a push notification for a coupon delivered to your phone for the very store you’re in. These types of personalized, location-based ads are exactly what advertisers and store owners can deliver. This is thanks to enhancements found in Bluetooth 5.0 along with Bluetooth Beacons’ broadcasting technology.
BLE and the Internet of Things (IoT)
The Internet of Things is the network of billions of devices – “things” – that are connected wirelessly to exchange data over the internet.
IoT technology helps to connect things like thermostats, smart lights,
people with embedded medical devices,
animals with implanted trackers,
and cars with driver-assist sensors.
Any person, animal, or man-made object can become a “thing” in IoT.
This means IoT is essentially connecting people, processes, and systems.
To make all of these connections possible, IoT relies on wireless technologies. ZigBee, Bluetooth Classic, and WiFi can all be used to connect devices wirelessly. However, BLE is often viewed as the most optimal technology for IoT applications because of two main reasons:
Low power consumption. Many IoT devices are battery-powered and need to last in the field for a very long time. For example, it would simply be impractical to regularly replace the batteries in hundreds of sensors in a warehouse environment.
The type of data being exchanged. BLE is optimized to transmit a small amount of data. This works great for IoT devices like sensors that just need to transfer state data.
The PCR test...
https://www.sec.gov/Archives/edgar/data/1403570/000149315221010243/form10-q.htm#V_009
Major Market Segments
Life Sciences.
The life sciences industry was one of the early areas of adoption of QD (Quantum Dots) technology, especially for QDs used in fluorescent markers in diagnostic applications. This includes both the in vitro use of QDs for marking (illuminating) particular cell types or metabolic processes for understanding diseases, and in vivo imaging made possible by QD fluorescence in near infrared that can be detected in deep tissues. The fluorescent qualities of QDs provide an attractive alternative to traditional organic dyes in bio-imaging. It is estimated that QDs are 20 times brighter and 100 times more stable than standard fluorescent indicators. QD technology is also being used in place of colloidal gold nanoparticles in lateral flow test kits such as those used in the rapid Covid 19 antigen test. QDs have been reported in literature to exponential improve the sensitivity of these test enabling earlier detection.
The Federal Drug Administration (“FDA”) has issued emergency use authorization (“EUA”) for medical tests that diagnose Covid-19.
The FDA is responsible for protecting the public health by ensuring safety, efficacy, and security of all human and veterinary drugs, biological products, and medical devices. With regards to medical tests, the FDA usually does this by making manufacturers meet rigorous guidelines in an approval process that can take many months. During an emergency, such as a pandemic, it may not be possible to have all the evidence that the FDA would usually have before approving a medical test. If there’s evidence that strongly suggests that patients have benefited from a test, the agency can issue an EUA to make it available.
One of the minimum requirements for granting EUA is that the known and potential benefits of the test outweigh the known potential risks.
However, this is a minimum requirement and not the standard. The minimum standard can be met and EUA is still not given; there may be additional requirements, such as the test meeting reasonable thresholds for safety and effectiveness and/or people in urgent need of care based on a diagnosis. EUAs are only given during a declared emergency; outside of this, an EUA is never given.
Once the pandemic is over and should FDA EUA of Covid-19 tests be revoked. The 510K approval process which requires validation and submission of the test for FDA 510(k) clearance, which is one of the normally used medical device regulatory pathways for FDA approval would be required to continue to sell the test kits in the USA.
It is important to note that QMC is collaborating with a leading university in the medical field and intends to initially pursue the FDA EUA while also pursuing the FDA 510(k) clearance of its test platform for non-covid related testing. QMC also intends to pursue regulatory approvals in one or more foreign jurisdictions.
The time required to complete either the FDA EUA or the FDA 510(k) approval process can vary widely, and approval is not guaranteed the same holds true for regulatory approvals outside the USA.
In 2019 the Company developed the QDX Ledger, which is based on technology acquired the blockchain-based technology assets of Capstan Platform, Inc. to provide an immutable, scalable and shared data store for consistent tracking and visibility among participants in a product supply chain. The identity and access credentials of participants, be they individuals, corporations or machines is also secured on the platform, providing mechanisms to control and restrict the supply of products as might be required by regulatory mandates and socially conscious business practices.
QDX Quantum Dots can be incorporated into almost any physical product so that its authenticity can be verified and tracked from point of manufacture through to sale to an end customer. Unlike existing approaches to establishing product identity, including QR code stickers and RFID tags, we believe that QDX Quantum Dots are more tamper proof, resistant to environmental extremes and low cost. We believe that they may be incorporated into products as diverse as auto parts, consumer electronics, apparel and luxury fashion accessories, industrial IoT devices, bank notes and even liquids, such as gasoline and lubricants.
In early 2020 and in the advent of the Covid 19 pandemic, the company recognized a need for a secure method for the validation and reporting of the Covid 19 testing process. The Company leveraged its existing QDX Ledger platform technology to launch the QDX HealthID later rebranded the QMC HealthID and is operated as a wholly owned subsidiary of Quantum Materials Corp.
Also, on January 26, 2021, the Company and Pasaca entered into a Distribution Agreement (the “Distribution Agreement”). Pursuant to the terms of the Distribution Agreement, the Company appointed Pasaca to act as an independent distributor to resell and distribute the Company’s Quantum Dots and QMC HealthID products.
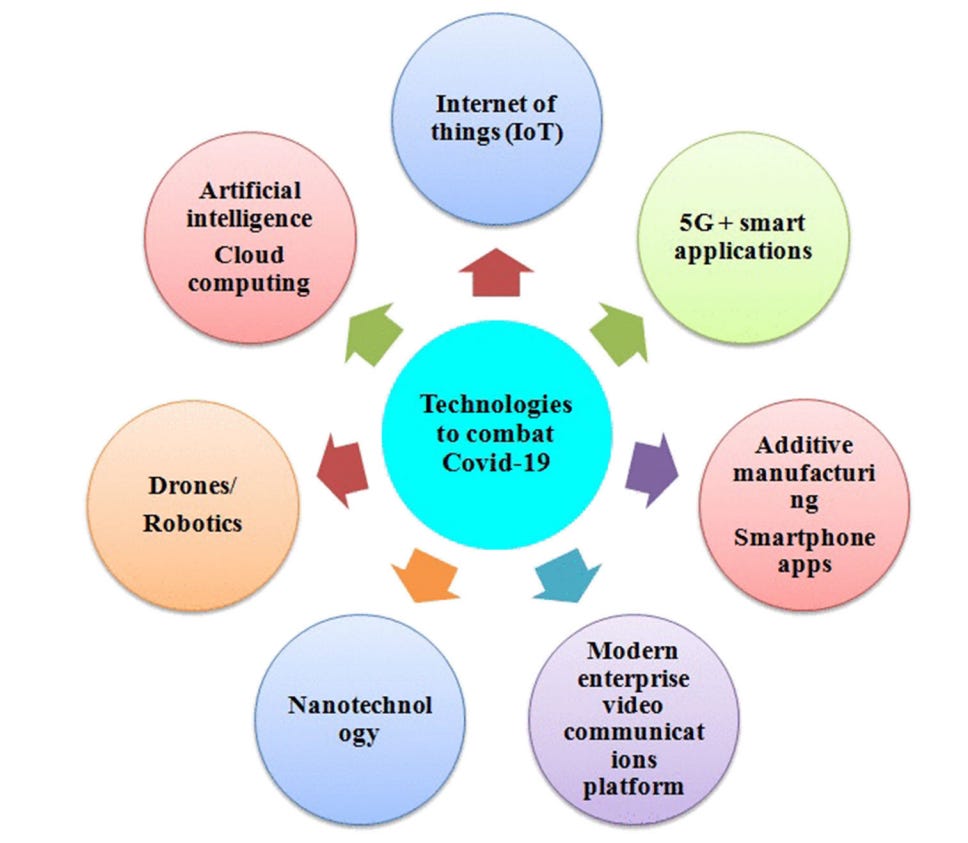
The pandemic of nanotechnology
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7883336/pdf/42247_2021_Article_178.pdf Nanotechnology: an emerging approach to combat COVID-19 (nih.gov)
Nanotechnology is science, engineering, and technology dealing at the nanoscale level.
It has made possible the development of nanomaterials, nano-biosensors, nanodrugs, and vaccines for diagnosis, therapy, and prevention of COVID-19.
https://phys.org/news/2023-11-networking-nano-biosensors-wireless-communication-blood.html
First, there was the Internet of Things (IoT) and now, at the interface of computer science and biology, the Internet of Bio-Nano Things (IoBNT) promises to revolutionize medicine and health care. The IoBNT refers to biosensors that collect and process data, nano-scale Labs-on-a-Chip that run medical tests inside the body, the use of bacteria to design biological nano-machines that can detect pathogens, and nano-robots that swim through the bloodstream to perform targeted drug delivery and treatment.
Nanoparticles
Small particles containing a few hundred atoms and size measured in nano-units are called nanoparticles (NPs).
These polymeric particles can either originate naturally or engineered by man. These are synthesized by various methods such as spinning, laser pyrolysis, molecular condensation, biological synthesis, mechanical milling, chemical etching, sputtering, and electro-explosion which are considered under bottom-up and top-down synthesis approaches [13]. NPs find their applications in various diverse fields like engineering, medicine, biotechnology, and pharmacology [14].
These particles are composed of three layers; the surface layer is functionalized with different molecules such as metals ions, polymers, and surfactants.
This small size accounts for most of the wondrous properties of nanoparticles. In their free state, nanoparticles show rapid mobility.
They absorb large amounts of solar energy and exhibit quantum effects.
NPs are available in various forms like carbon-based nanoparticles, metal nanoparticles, and lipid-based nanoparticles.
Carbon nanotubes (CNTs) and fullerenes are two main classes of carbon-based NPs [16].
CNTs generally have tubular and elongated structure 1–2 nm in diameter [17]. These are similar to a graphite sheet roll but may range from single- to multi-walled carbon nanotubes. They are extensively synthesized by deposition of carbon precursors and chemical vapor and finally used in commercial applications such as gas adsorbents and fillers [18].
Applications of nanomaterials
Smartphone-assisted sensing
Smartphone-based sensing systems are the developing systems with semi-automated user interface and the accessibility due to less required knowledge and training. By the proper usage of the hardware and software, sensing systems can be developed with smartphones and can be used by normal people. There are numerous ways to connect a peripheral module to the phone. A sensing system can be connected to smartphones by proprietary interfaces, USB communication and power, audio headphone port, Bluetooth, and NFC [42].
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7474418/ Wireless Technologies for Implantable Devices - PMC (nih.gov)
An implantable sensor is a type of implantable device that contains one or more sensing elements, such as strain gauges or pressure sensors, which perform localized measurements. One of the advantages of wireless implantable devices is that they do not need physical wires to interface with external technology. Compared to wired sensors, the wireless nature of implantable sensors leads to a different set of design considerations regarding communication and power. For example, wireless communication in tissue introduces unique challenges that are not present in wireless technologies outside the body. Biological tissues significantly attenuate wireless signals, especially at commonly used frequencies like 2.4 GHz. Safety is an additional concern because there is a need to limit the absorption of damaging energy into the tissue.
It is common to use technologies that are designed for ex vivo use like Bluetooth, but these solutions are rarely ideal.
Several alternative methods were researched for implantable devices [6,7], but they are not currently widely used.
Many researchers and sensor developers use existing communication technologies for implantable sensors such as WiFi, Bluetooth, and ZigBee [19,20].
Bluetooth is the technology of choice for a device that is designed to connect with a device like a mobile phone or computer, and pairing to these devices is straightforward, while BLE (Bluetooth Low Energy) builds on that foundation with power-saving innovations for wireless sensors.
Zigbee is associated with mesh networks containing many devices that can connect to any other device in the network, which may be convenient for a system containing several devices on the body.
Safety factors are important when selecting the frequency band. The Federal Communications Commission (FCC) defines two categories of electromagnetic radiation: ionizing and non-ionizing [25].
Ionizing radiation, which causes changes to biological tissue on the molecular level, is caused by radiation at frequencies in and above the ultraviolet range.
Wireless transmission through human body also needs to maintain a safe level of electromagnetic energy to prevent tissue heating and meet safety standards.
While there are a number of ways to passively interrogate sensors from a distance, the most common methods that are practical for implantable devices are based on electromagnetic (EM) fields (typically in radiofrequency range) [36], magnetic energy [37], and acoustic energy [38]. Electromagnetic-based passive sensors are typically made of a simple electrical resonance circuit consisting of an electrical inductor (L), capacitor (C), and resistor (R). When exposed to the EM fields, these sensors undergo resonance when it emits the highest returned signal. The parameters are typically tracked by measuring the change in the resonance frequency. In addition to the basic resonance circuit sensors that use the RLC components [36], there are a few different variations of EM-based passive sensors, such as RFID sensors [39], which have microchips that can perform more complex sensing functions, a varactor-based LC sensor that can convert voltage change to resonance frequency shifts [40], and LC sensor arrays that can measure multiple parameters simultaneously [41].
Ultrasound energy provides another means to remotely power implantable sensors.
Similar to EM-based sensors, magnetic-based sensors use a magnetic field to provide the energy needed for internal measurement functions and wireless communication.
One promising method for energy harvesting is the use of photovoltaic power (Table 3). Near-infrared (NIR) light present in sunlight can pass through skin and tissue with low power losses [71].
Various subcutaneous solar panels were proposed for biomedical implants [55,72]
Quantum Materials Corp. announces a high volume production process for perovskite quantum dots
Concerns
Toxicity
FALSE POSITIVE in efficiency measurements
👆👆👆👆👆👆👆👆👆👆👆👆👆👆👆👆👆👆
FALSE POSITIVE ❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗
❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗
https://web.archive.org/web/20191228095141/https://www.quantummaterialscorp.com/
In this article, we’ll highlight how the combination of blockchain and nanotechnology can be applied to a particularly challenging aspect of supply chain management, namely the huge global criminal marketplace in counterfeit goods, which hurts business profits, impacts brand trust and undermines customer relationships.
At Quantum Materials Corp. we have developed nanomaterials called quantum dots over the past decade. Quantum dots are nanoscale semiconductor particles that possess notable and extremely useful optical and electrical properties. They measure from 1,000 to 100,000 atoms in size (approximately 10,000 dots would fit across the diameter of a human hair) and they generate light when energy is applied to them or generate energy when light is applied.
QMC creates in commercial quantities quantum dots that can be finely tuned to emit predetermined wavelengths of light (in both the visible and non-visible spectrums) with the ability to create billions of unique optical signatures. Moreover, they are excitable by numerous excitation energy sources.
Our quantum dots can be incorporated into almost any physical item at time of manufacture, and then provide a unique light signature that establishes absolute product identity. These identities are impossible to copy or clone so that products enhanced by them can be verified as being genuine items and not counterfeits.
When the quantum dot signature of a product is scanned (via a hand-held scanner or an app on a smartphone), a digital representation is created that is stored on our secure and tamper-proof blockchain platform. It is this platform that allows for tracking of products providing visibility among all participants in their supply chain - from manufacture to customer purchase.
In addition, the blockchain platform is also used to store the unique digital identities of individual customers, and to tie ownership of a product to a customer at purchase time. No longer is it necessary to keep the receipt!
👆👆👆👆👆👆👆👆👆👆👆👆👆👆👆👆👆👆
THIS IS WHAT DIGITAL IDENTITY IS ALL ABOUT!!!
❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗❗
"At least these Smart Cards gives you something to read whilst you're waiting 2 hours for the next bus."
Nanotechnology is improving smart cards. A smart card has
a microchip in it which makes it 'smart'.
It provides not only memory capacity, but computing capability as well and thus the chip is capable of processing data.
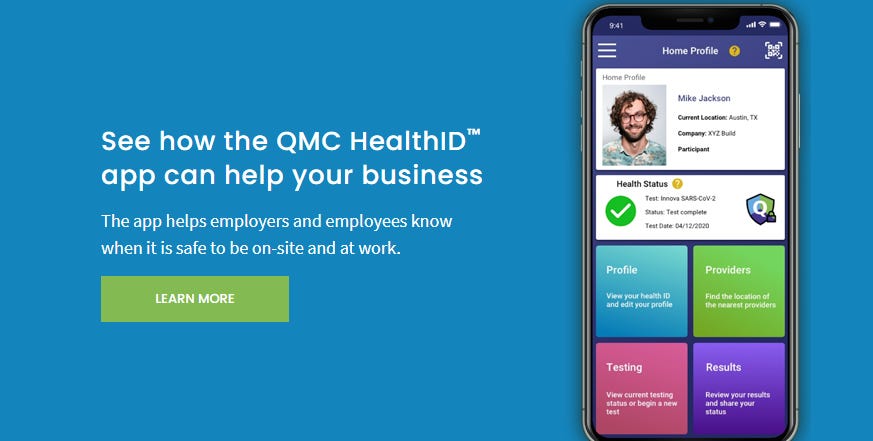
https://web.archive.org/web/20201225164329/https://www.qmchealthid.com/ QMC HealthID - Control and Share your Health Status Information App (archive.org)
HealthID
We have expended considerable resources developing and marketing our HealthID product but have not yet achieved the anticipated success. Despite the substantial interest and verbal commitments, primarily from U.K.-centric companies, the wide-ranging extended shut down in the U.K. interfered with our ability to get the product adopted. Although we still believe QMC HealthID has great potential, we also think it has much more synergy with our quantum dot point of care testing technologies. We will also be seeking out potential strategic partners as well as potential acquirers for this platform. As a result, this product will not be part of our immediate commercialization efforts.
DOT-IQ, point of care testing technology
The Company recognized early in the advent of Covid-19 that we could use our proprietary technologies to design and develop a more sensitive lateral flow test kit using our unique quantum dot technologies. Over the past several months, we have expanded our biotech team and collaborated with our material scientists. As a result, we have now developed a point of care test platform (POCT) that we believe is more sensitive than current technologies. We believe it will provide analytic sensitivity that can rival PCR and provide quantitative results when warranted. We are currently developing 1) LFTs (lateral flow test kits) for TBI (traumatic brain injury, aka concussions), 2) a multiplex test that detects Covid-19, Flu and RSV (Respiratory Syncytial Virus), and 3) a vaccine validator to thwart counterfeit vaccines and validate vaccine efficacy. This test kit technology uses a unique quantum dot as a signal amplifier and can give a visual test result indication using a low-cost UV light source or using our POCT reader to interface with a P.C. or a smartphone. This reader is designed to provide a binary readout indicating positive or negative and a quantitative result indicating the level of viral loading. We believe that this can be very valuable, particularly in viruses like Covid-19, where a current test cannot reliably identify low viral loading, which is often the case in asymptomatic individuals who are the most likely to spread the virus unknowingly. In addition, the Company has signed a term sheet with a Canada-based biotech company that has identified and has a patent-pending on several biomarker candidates that will be used to develop the TBI POCT. The term sheet is non-binding and we can provide no assurance that we will be able to reach definitive agreements on terms acceptable to the Company. We are in the process of negotiating the final terms that include the payment milestones required for QMC to design, develop, shepherd the FDA approval process, and produce the test kits.
















I'm glad I don't know the answer to your last question.
I do know this,
Where there is kindness there is goodness.
Where there is goodness there is magic.
Good night, I appreciate you.
Thank you for sharing and expanding upon the topic.